The MDCG has released a very important guidance on the ”device type” to be shown on the implant card of an implantable medical device as per the MDCG Guidance 2019-8 issued under Article 18 of the MDR that obliges:
1. The manufacturer of an implantable device shall provide together with the device the following:
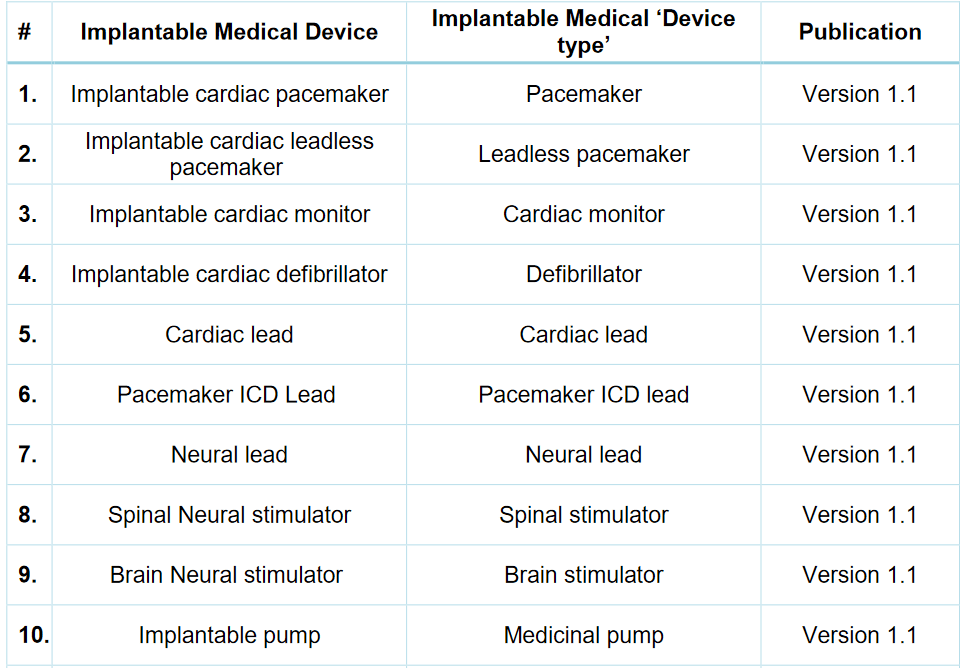
The MDCG 2019-8 requires from a manufacturer of an implantable medical device to include a ”device type” also as information allowing the identification of a particular device (see below highlighted in red). The new Guidance presents a non-exhaustive list of implantable medical ‘device types’ to assist manufacturers in generating the implant card of their devices.
Source: MDCG
Currently, the Guidance includes 88 ”device types”, subject to further updates. Below, an extract of the first 10 entries.
Source: MDCG
This new MDCG Guidance as well 2019-8 V2 Guidance are available in our Library of documents –in MDCG category.
Do not forget to follow us on LinkedIn!



Leave a Reply