The Medical Devices Coordination Group (MDCG) has published a guidance document on Safety reporting in the context of clinical investigations.
According to Article 80(2) and Article 73 of the MDR and without delay to all Member States in which the clinical investigation is being conducted, safety reporting should include all of the following:
- any serious adverse event that has a causal relationship with the investigational device, the comparator or the investigation procedure or where such causal relationship is reasonably possible;
- any device deficiency that might have led to a serious adverse event if appropriate action had not been taken, intervention had not occurred, or circumstances had been less fortunate;
- any new findings in relation to any event referred to in points a) and b).
The Guidance particularly addresses the Safety reporting obligation towards EUDAMED in absence of the electronic system referred to in Article 73 being available at the moment of the entry into force of the MDR.
Safety reporting guidance is provided for both Pre-market clinical investigations and for Post-Market Clinical Follow Up (PMCF) investigations.
Drug Trials are also addressed in Section 2.2.
Definitions relevant to this procedure are outlined in Section 3, while Section 4 gives an overview of the possible reporting methods, together with a useful table, that sums up the possible formats to use and when to use them (the table can be found here, together with the full text of the Guidance):
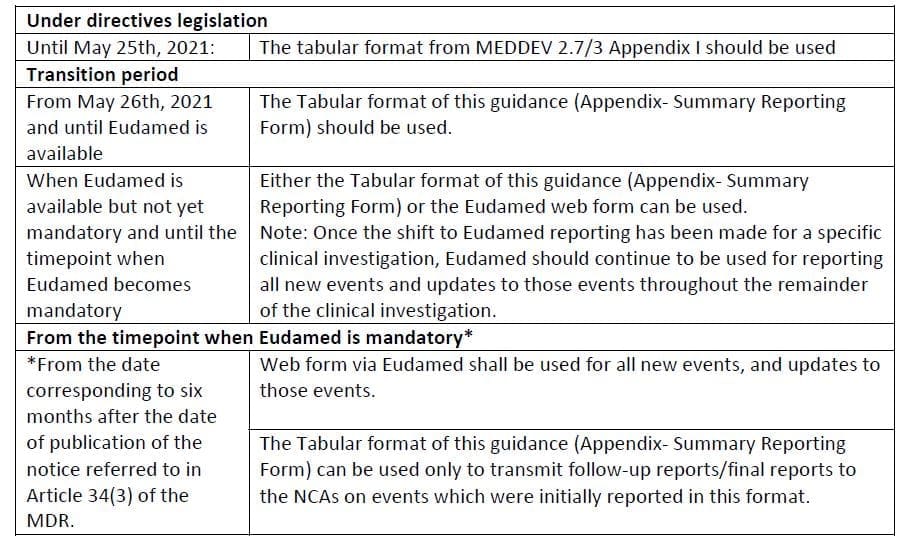
The Summary Reporting Form mentioned above is available as Appendix to the Guidance.
A list of the events that need to be reported by the sponsor of the clinical investigation, which could be the manufacturer, the legal representative or another person or entity to NCAs, where the clinical investigation has commenced is presented in Section 5.
Section 8 and 9 respectively address the timelines and the assessment of the causal relationship between the use of the medical device and each adverse event.
We will keep you informed about any further update on our News section .
More information on Clinical investigations can be found on MDlaw Library of Documents.


Leave a Reply