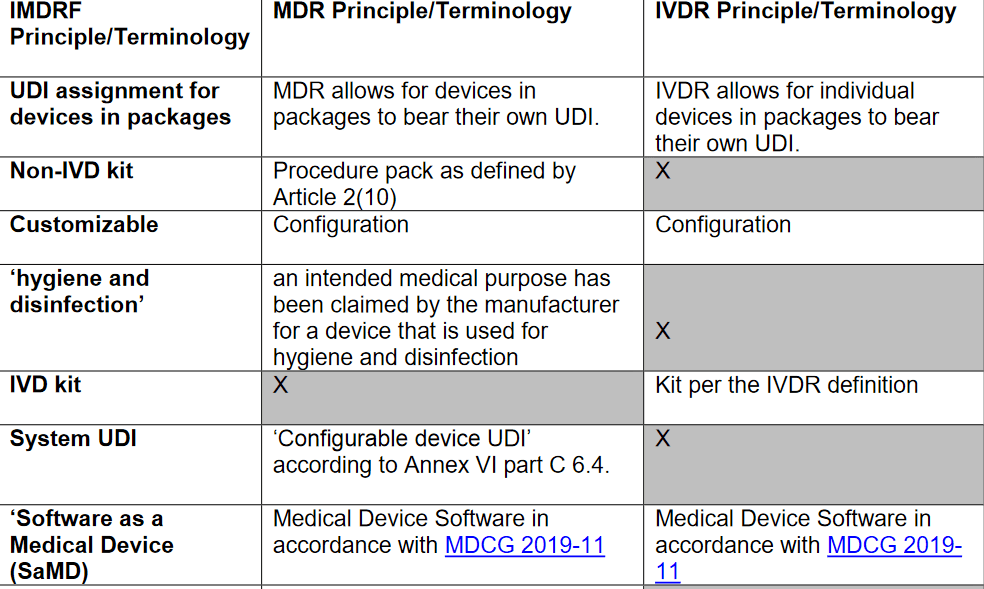
The MDCG has endorsed two new documents today! The first FAQ MDCG 2021-12 lays down questions and answers on the EMDN (European Medical Device Nomenclature) and the MDCG 2021-10 provides clarifications on how certain principles and examples from IMDRF N48 Appendixes E-I (UDI Application Guide) apply under the MDR/IVDR, including a comparison table that is non-exhaustive and underlines that the document should be read together with the legal texts of EU MDR & IVDR. Below, an extract of the MDCG comparison table:
Moving to the FAQ on the EMDN, the MDCG replies to the below 5 questions, where it is also indicated that the second version of EMDN will be released in Q3 2021, in addition to the new terms and descriptions for medical device software.
- What is the EMND?
- How was the EMDN created?
- What are the key principles of EMDN?
- How do I gain access to the EMDN?
- How is the EMDN structured?
- Which level of the EMDN should I use to assign a term to my device?
You can find both documents in our Library of Documents and its dedicated categories on MDCG, UDI, EUDAMED – under MDR and IVDR!


Leave a Reply