The MDCG has endorsed a 2021-21 Guidance on performance evaluation of SARS–CoV–2 in vitro diagnostic medical devices for the conformity assessment under IVDD and IVDR. The following devices are covered: IVDs for detection or quantification of SARS-CoV-2 nucleic acid, antigens and also detection or quantification of antibodies against SARS-CoV-2.
It is especially important to read through the Guidance, as it will present the basis for future Common specifications under the IVDR, which should be adopted in the coming months.
The newest MDCG Guidance firstly provides some relevant definitions, e.g. what is LOD, analytical specificity, 95% positive cut–off value for NAT assays and clarifies some Overall considerations related to performance evaluations:
- They should be done with direct comparison with the state-of the-art device;
- The device used for comparison should be one bearing CE marking, if on the market at the time of the performance evaluation;
- For anti-SARS-CoV-2 tests, the new device should have an overall performance at least equivalent to that of the state of the art device of the same type;
- Devices used for determination of status of samples used in performance evaluations of SARS–CoV–2 IVDs should be state–of–the–art devices bearing CE marking;
- They should be performed on a population equivalent to the European population;
- Discrepant results should be resolved as far as possible by for ex. use of an alternative method or marker, a review of the clinical status and diagnosis of the patient;
- As part of the risk analysis, the whole system failure rate leading to false–negative results should be determined in repeat assays on low–positive specimens.
Moreover, the following aspects are addressed:
Sensitivity and specificity: E.g. Positive specimens used in the performance evaluation should be selected to reflect different stages of the respective disease(s), different antibody patterns, different genotypes, different subtypes, mutants, etc. Negative specimens used in a performance evaluation should be defined so as to reflect the target population for which the device is intended, such as blood donors, hospitalized patients, pregnant women, etc. Specificity should be calculated using the frequency of repeatedly reactive (i.e. false positive) results in individuals negative for the target marker (MDCG, 2021).
Interference and cross-reactivity: E.g. The manufacturer should select the potential interfering substances to be evaluated taking account of the composition of the reagents and configuration of the device. The manufacturer should include specimens such as, where applicable: those representing related infections; those from multipara; those containing human antibodies to components of the expression system (MDCG, 2021).
Anticoagulants: The performance evaluation should verify the performance of the device using all anticoagulants which the manufacturer indicates for use with the device – for COVID-19 IVD intended for use with plasma. This should be demonstrated for at least 50 plasma specimens per anticoagulant (25 positive and 25 negative). (MDCG, 2021).
Batch testing: The manufacturer’s batch testing criteria should ensure that every batch consistently identifies the relevant antigens, epitopes and antibodies, and is suitable for the claimed specimen types (MDCG, 2021) – for COVID-19 antigen and antibody IVDs.
Self–tests: E.g. Self-testing IVDs should meet the same requirements for sensitivity and specificity as respective devices for professional use. The lay person selected for the performance evaluation should be representative of the intended users groups (MDCG, 2021).
Finally, the MDCG presents 7 tables for the following IVD types with specific considerations.
- First–line assays (including rapid tests) for antibodies against SARS–CoV–2 (anti–SARS–CoV–2): IgG–only, IgG combined with IgM and/or IgA, and total antibody.
- Assays (including rapid tests) for anti–SARS–CoV–2: IgM and/or IgA detection.
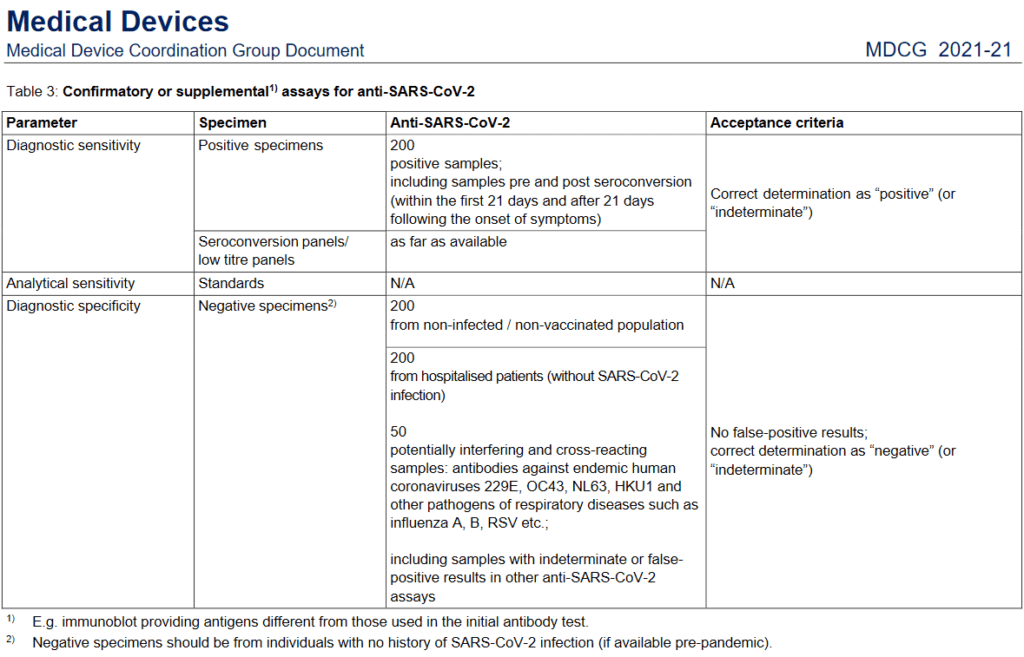
- Confirmatory or supplemental assays for anti–SARS–CoV–2.
- Antigen assays (including rapid antigen tests) for SARS–CoV–2.
- NAT assays for SARS–CoV–2 RNA.
- Additional requirements for SARS–CoV–2 antigen Self–Tests.
- Additional requirements for SARS–CoV–2 antibody Self–Tests.
6. and 7. are intended for devices which have already undergone a performance evaluation for professional use.
Image 1: Table 3 Confirmatory or supplemental assays for anti-SARS-CoV-2.
Source: MDCG, 2021
You can find the full text of the Guidance in our dedicated IVDR Library – MDCG


Leave a Reply