Legacy devices are devices that are typically covered by a valid CE Certificate or are being up-classified by the Regulations and benefit from a special status under the Medical Devices Regulation 2017/745 (MDR) and the In Vitro Diagnostic Devices Regulation 2017/746 (IVDR).
The purpose of this status was to allow these devices to be placed on the EU market after MDR/IVDR date of application, while the manufacturers implement the new requirements and the applications for an MDR/IVDR CE Certificate are processed.
MDR
On 20 March 2023, manufacturers of MDR legacy devices were granted an extension of the transitional period to the following dates:
- 26 May 2026 for class III implantable custom-made devices, provided the manufacturer has applied for a conformity assessment before 26 May 2024.
- 31 December 2027 for class III and class IIb implantable devices, except sutures, staples, dental fillings, dental braces, tooth crowns, screws, wedges, plates, wires, pins, clips and connectors.
- 31 December 2028 for class IIb devices other than those mentioned above, Class IIa devices, Class I devices placed on the market in sterile condition or having a measuring function, and class I devices that will need to involve a notified body for the MDR conformity assessment (up-classified devices).
Nevertheless, to benefit from this extended period, manufacturers must meet extra requirements as per the Regulation 2023/607:
- Devices do not present an unacceptable risk to the health or safety of patients.
- No significant changes in the design and intended purpose of the devices.
- Manufacturer has set up an MDR complaint QMS by 26 May 2024 (Article 10.9 MDR).
- Have a look at our article, where we presented you with necessary elements to achieve complaint QMS.
- Manufacturer is compliant with MDD and the legacy requirements set in the MDR. Relevant MDlaw posts:
- MDCG guidance on NB surveillance & QMS
- PMCF Templates, Clinical evidence for legacy devices
- EUDAMED Management of Legacy Devices & registration
- Manufacturer or his AR has applied to a notified body for a conformity assessment under the MDR – by the latest 26 May 2024;
- Manufacturers must approach a Notified Body to assess their conformity with the MD Regulations early enough to issue the MDR CE Certificates. As shown in the European Commission’s latest survey on Notified Bodies, as of March 2023, only 2951 MDR CE Certificates and 331 IVDR CE Certificates were issued. Furthermore, manufacturers struggle to find Notified Bodies whose designation activities are all within the scope of their device applications, as this is one of the main reasons for the refusal of MDR & IVDR applications.
- It is key for manufacturers of legacy devices to not only apply to a Notified Body as soon as possible, but also to apply to the correct one.
- Manufacturer and notified body have signed a written agreement – by the latest 26 September 2024.
MD Legacy timeline
IVDR (Update: July 2024)
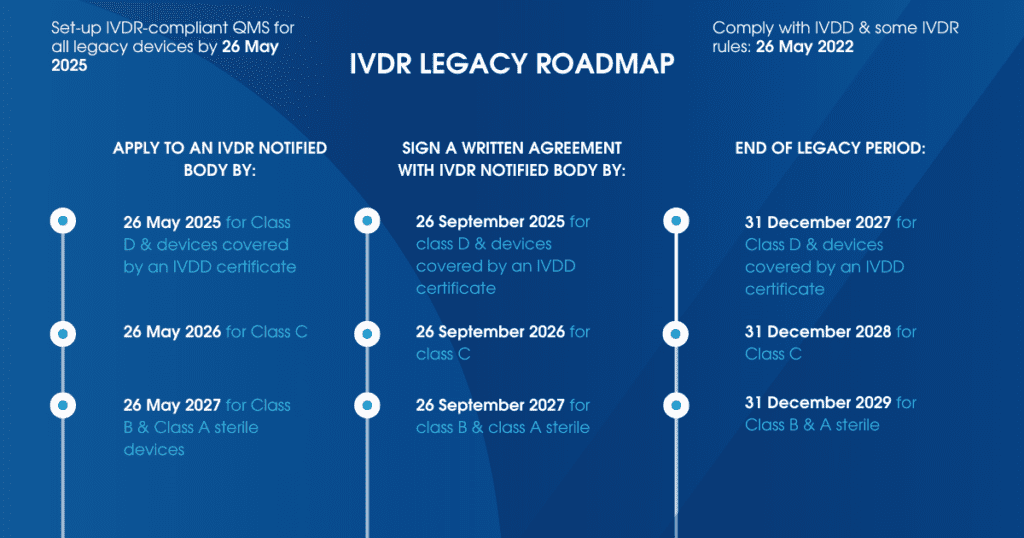
On 9 July 2024, manufacturers of IVDR legacy devices were granted an extension of the transitional period to the following dates:
- 31 December 2027 for IVD up-classified to Class D under IVDR and devices covered by an IVDD CE Certificate
- 31 December 2028 for IVD up-classified to Class C under IVDR
- 31 December 2029 for IVD up-classified to Class B and A sterile under IVDR
To benefit from this extended period, manufacturers must meet extra requirements outlined in the Regulation 2024/1860:
- Devices do not present an unacceptable risk to the health or safety of patients.
- No significant changes in the design and intended purpose of the devices.
- Manufacturer has set up an IVDR complaint QMS by 26 May 2025 (Article 10.8 IVDR).
- Have a look at our article, where we presented you with necessary elements to achieve complaint QMS or contact Obelis for its dedicated service on ISO 13485.
- Manufacturer is compliant with IVDD and the legacy requirements set in the IVDR. Relevant MDlaw posts:
- MDCG Guidance on IVDR legacy requirements
- MDCG guidance on NB surveillance & QMS
- PMCF Templates that could be used under IVDR
- Manufacturer or his AR has applied to a notified body for a conformity assessment under the IVDR:
-
- by the latest 26 May 2025 for IVD up-classified to Class D under IVDR and devices covered by an IVDD CE Certificate
- by the latest 26 May 2026 for IVD up-classified to Class C under IVDR
- by the latest 26 May 2027 for IVD up-classified to Class B and A sterile under IVDR
-
- Manufacturer and notified body have signed a written agreement
-
- by the latest 26 September 2025 for IVD up-classified to Class D under IVDR and devices covered by an IVDD CE Certificate
- by the latest 26 September 2026 for IVD up-classified to Class C under IVDR
- by the latest 26 September 2027 for IVD up-classified to Class B and A sterile under IVDR
-
- Only for manufacturers of a device covered by a Directive’s (IVDD) Certificate: the Certificate must be valid on 9 July 2024 (date the new Regulation was published in the Official Journal). If the IVDD Certificate has expired before 9 July 2024, the manufacturer has signed an agreement with a notified body before the expiry date of the Certificate or the manufacturer has obtained a national derogation measure (e.g., Article 54 IVDR).
Manufacturers must approach a Notified Body to assess their conformity with the IVD Regulations early enough to issue the IVDR CE Certificates.
It is key for manufacturers of legacy devices to not only apply to a Notified Body as soon as possible, but also to apply to the correct one.
Obelis can assist you to select the Notified Body that most adapts to your needs.


Leave a Reply